獨克士黴素
此條目需要擴充。 (2010年9月19日) |
 | |
 | |
| 臨床資料 | |
|---|---|
| 讀音 | /ˌdɒksɪˈsaɪkliːn/ DOKS-iss-EYE-kleen |
| 商品名 | Doxy、Doryx、Vibramycin及其他 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682063 |
| 核准狀況 | |
| 懷孕分級 |
|
| 給藥途徑 | 口服給藥, 靜脈注射[1] |
| ATC碼 | |
| 法律規範狀態 | |
| 法律規範 |
|
| 藥物動力學數據 | |
| 生物利用度 | ~100% |
| 血漿蛋白結合率 | 80–90% |
| 藥物代謝 | 微小,可忽略不計 |
| 生物半衰期 | 10–22小時 |
| 排泄途徑 | 多數透過糞便, 40%透過尿液 |
| 識別資訊 | |
| |
| CAS號 | 564-25-0 |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.008.429 |
| 化學資訊 | |
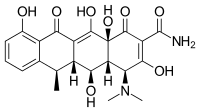
| 化學式 | C22H24N2O8 |
| 摩爾質量 | 444.44 g·mol−1 |
| 3D模型(JSmol) | |
| |
| |
獨克士黴素(INN:doxycycline,也稱為多喜黴素、強力黴素、偉霸黴素、去氧土黴素或去氧土黴素)是種廣譜四環素類抗生素,用於治療病原細菌和某些寄生蟲的感染[1] - 如細菌性肺炎、痤瘡、披衣菌感染、萊姆病、霍亂、斑疹傷寒和梅毒,[1]也可用於預防瘧疾。[2][3]
使用後常見的副作用有腹瀉、噁心、嘔吐、腹痛以及曬傷風險增加。[1]不建議個體在懷孕期間使用,以免對胎兒造成傷害。[1]此藥物與其他四環素類抗生素藥劑一樣,係透過抑制病菌蛋白質的生成而減緩其繁殖或是將其殺死。[1][4]它透過靶向瘧原蟲的色素體胞器(頂質體),而將其殺死。[5][6]
獨克士黴素於1957年取得專利,並於1967年由輝瑞製藥取得醫療用途核准。[7][8]它已納入世界衛生組織基本藥物標準清單之中。[9]市面上有其通用名藥物流通。[1][10]此藥物在美國2022年最常使用處方藥中排名第68,開立的處方箋數量超過900萬張。[11][12]
醫療用途
[編輯]

獨克士黴素除能用於四環素類抗生素所有成員的一般適應症外,也常用於治療萊姆病、慢性前列腺炎、鼻竇炎、骨盆腔發炎、[13][14]嚴重痤瘡、酒糟鼻、[15][16][17]和立克次體感染。[18]口服此藥物以治療丘疹膿皰性酒糟鼻和成人痤瘡的藥理作用不僅限於其抗菌活性,它的抗炎作用和對血管生成抑制作用也是療效的重點。[19]
此藥物於2004年在加拿大被認為是治療披衣菌感染和非淋菌性尿道炎的一線藥物,並與頭孢克肟聯合用於治療無併發症的淋病。[20]
抗菌性
[編輯]一般適應症
[編輯]獨克士黴素是一種廣譜抗生素,用於治療多種細菌感染。對卡他莫拉氏菌、羊布魯氏菌、肺炎披衣菌、肺炎支原體等病菌能發揮作用。此外此藥物也用於預防和治療炭疽、鈎端螺旋體病、腺鼠疫和萊姆病等嚴重疾病。然而一些細菌,包括嗜血桿菌屬、人類支原體和綠膿桿菌,已出現對獨克士黴素的抗藥性。[21][22]此藥物也能有效對抗鼠疫耶爾森菌(腺鼠疫的傳染源),並用於治療萊姆病、[23][24][25][26]埃利希體病、[27][28]和落磯山斑點熱。[29]
- 落磯山斑點熱、斑疹傷寒和斑疹傷寒群(一組由立克次體引起的傳染病)、恙蟲病、[31]Q型流感、[32]立克次氏體痘和由立克次體引起的蜱熱、[33][34][35]
- 肺炎枝原體引起的呼吸道感染、[36]
- 花柳性淋巴肉芽腫、沙眼、結膜炎及砂眼衣原體導致的成人單純性尿道、子宮頸內膜或直腸感染、[29][30]
- 鸚鵡熱,[29][30]
- 解脲脲原體引起的非淋菌性尿道炎、[29][30]
- 伯氏疏螺旋體引起的回歸熱、[29][30]
- 杜克雷嗜血桿菌引起的軟性下疳、[29][30]
- 鼠疫耶爾森菌引起的腺鼠疫、[29][30]
- 兔熱病、[29][30]
- 霍亂、[29][30]
- 彎曲桿菌胎兒型感染、[29][30]
- 布魯氏菌屬引起的布魯氏菌病(與鏈黴素聯合使用)、[29][30]
- 巴爾通體病、[29][30]
- 腹股溝肉芽腫(克雷伯氏菌屬)、[29][30]
- 萊姆病:[37]可用於成人及兒童,且適用於各年齡層兒童的治療或預防,最長治療週期為21天。[38]
特定革蘭氏陰性菌適應症
[編輯]若細菌學檢測證實感染病原為對獨克士黴素敏感的革蘭氏陰性菌,則可選用此藥物進行治療:[29][30]
- 大腸桿菌感染、[29][30]
- 產氣腸桿菌感染、[29][30]
- 志賀氏菌屬感染、[29][30]
- 不動桿菌屬感染、[29][30]
- 流感嗜血桿菌引起的呼吸道感染、[29][30]
- 克雷伯氏菌屬引起的呼吸道感染和泌尿道感染。[29][30]
特定革蘭氏陽性菌適應症
[編輯]一些革蘭氏陽性菌已對獨克士黴素產生抗藥性。高達44%的化膿性鏈球菌和高達74%的糞鏈球菌屬已具抗藥性。高達57%的痤瘡丙酸桿菌菌株已具抗藥性。[39]若細菌學檢測證實感染病原為對獨克士黴素敏感的革蘭氏陽性菌,則可選用此藥物進行治療:[29][30]
當病人禁用青黴素時
[編輯]當個體不能施用青黴素時,可使用獨克士黴素治療:[29][30]
- 由梅毒密螺旋體引起的梅毒、[29][30]
- 由雅司螺旋體引起的熱帶肉芽腫、[29][30]
- 由單核細胞增生李斯特菌引起的李斯特菌病、[29][30]
- 由梭桿菌屬引起的壞死性牙齦炎、[29][30]
- 由衣氏放線菌引起的放線菌病、[29][30]
- 由梭菌屬引起的感染。[29][30]
用作輔助製劑
[編輯]獨克士黴素也可用作嚴重痤瘡的輔助製劑。[40][29][30]
亞抗菌劑量獨克士黴素 (SDD) 廣泛用作牙周炎刮治和根面平整術的輔助用藥。SDD也用於治療痤瘡和酒糟鼻等皮膚病,[15][41][42]包括眼部紅斑痤瘡。
此藥物也用作軟性下疳的輔助製劑。[43]
作為性傳染病預防用途
[編輯]此藥物可用於暴露後預防(PEP),以減少性傳染病感染(STI)的發生率,但它與相關的四環素抗藥性有關聯,特別是淋球菌。[44][45][46]澳大利亞共識聲明因而提到基於抗生素耐藥性的考量,獨克士黴素在暴露後預防中的應用應予謹慎。目前證據顯示此藥物對於男男性行為者梅毒的預防效果較佳,但對於其他細菌性性傳染病感染,其益處與風險的比值尚不明確。[47]
美國疾病管制與預防中心 (CDC)[48]和澳大利亞愛滋病醫學協會的指引支持在PEP中適當使用獨克士黴素。[49][50]
組合使用
[編輯]治療布魯氏菌病的一線藥物是獨克士黴素和鏈黴素的組合,二線的是獨克士黴素和利福平的組合。[51]
抗瘧藥
[編輯]獨克士黴素對惡性瘧原蟲的紅血球階段(erythrocytic stage)具有活性,但對惡性瘧原蟲的配子母細胞沒作用。[52]此藥物可用於預防瘧疾。不建議單獨用於瘧疾的初始治療,雖然寄生蟲會對此藥物的效力敏感,但其抗瘧作用會延遲發生,瘧原蟲在此期間可能繼續繁殖,有可能導致病情惡化。。[53]
獨克士黴素會阻斷惡性瘧原蟲頂質體(細胞器)中的蛋白質產生,而發生兩重要作用:破壞寄生蟲產生對其生長非常重要的脂肪酸能力,且損害瘧原蟲血紅素(一種輔助因子)的產生。這些影響發生在寄生蟲生命週期的後期,對寄生蟲的抑制作用主要發生在血液期,此階段是瘧原蟲引起宿主發病的主因。[54]獨克士黴素透過此過程,既能抑制惡性瘧原蟲的生長,又能防止其繁殖。它不會直接殺死惡性瘧原蟲,而是創造阻止其生長和複製的條件。[55]
世界衛生組織 (WHO) 發佈的指南指出,獨克士黴素與青蒿琥酯或是奎寧組合後,可用於治療惡性瘧原蟲引起的無併發症瘧疾,或作為嚴重瘧疾靜脈注射治療後的續接療法。[56]
驅蟲藥
[編輯]獨克士黴素可殺死線蟲生殖道中的共生沃爾巴克氏體屬細菌,導致線蟲不孕,而減少蟠尾絲蟲症和淋巴絲蟲病等疾病的傳播。[57]於2005年進行的現場試驗,顯示為期8週的獨克士黴素療程幾乎將微絲蚴的釋放清除。[58]
硬化劑注射療法
[編輯]獨克士黴素也用於輔助治療緩流血管畸形(即靜脈畸形和淋巴畸形),以及術後淋巴液瘤的血管硬化劑注射療法。[59]
仿單標示外使用
[編輯]獨克士黴素可用於仿單標示外用途,以治療家族性澱粉樣物多發性神經病變 (ATTR)。研究顯示獨克士黴素與熊去氧膽酸的聯合療法,可有效破壞ATTR患者體內已形成的轉甲狀腺素蛋白纖維,為治療此病提供新的希望。[60]
給藥途徑
[編輯]獨克士黴素可透過口服或是靜脈注射途徑給藥。[1]
此藥物與乳製品、抗酸劑、鈣補充劑、含鐵製劑、含鎂瀉藥或膽汁酸螯合劑一同服用並不危險,但這些食物和補充劑都可能會降低人體對獨克士黴素的吸收率。[61][62]
禁忌症
[編輯]有嚴重肝病或同時使用異維A酸或其他類視黃醇(與維生素A相似的有機化合物)禁用此藥物,因為在極少數情況下,四環素和類視黃醇均會導致高顱內壓。[61]
懷孕與哺乳
[編輯]獨克士黴素被FDA歸類為D級妊娠藥物(風險證據明確:已有明確證據顯示對人類胎兒有風險)。它會進入母乳。[63]其他四環素抗生素於個體在懷孕期間和8歲以下兒童禁用,因為它們可能會破壞骨骼和牙齒的發育。[64]
不良反應
[編輯]使用此藥物的通常不良反應有頭痛、噁心、嘔吐及皮膚對陽光敏感。[65] 嚴重副作用屬於罕見(少於0.1%):不明原因的流血,包括流鼻血(可能是血液問題)、腹瀉且便中有血或是粘液、耳鳴、淺色排便及深色尿液(可能是肝臟問題)、用藥後開始關節痛或肌肉痛、劇烈頭痛連同嘔吐及視力問題以及光照性指甲剝離等。[65]
藥物交互作用
[編輯]先前人們認為獨克士黴素會加速細胞色素P450(一個龐大的酶蛋白質超家族)分解避孕藥,而損害多種激素避孕藥的有效性。研究顯示使用大多數四環素類抗生素(包括獨克士黴素)時,口服避孕藥的有效性並無明顯下降,但許多醫生仍然建議服用此藥物者另採屏障避孕法以防意外懷孕。[66][67][68]
藥理學
[編輯]獨克士黴素與其他四環素抗生素同樣具有抑菌作用 - 透過抑制病菌的蛋白質合成來防止其繁殖。[69]
作用機轉
[編輯]獨克士黴素是一種廣譜抑菌抗生素。它透過與僅存在於病菌中的30S核醣體亞基結合來抑制其蛋白質合成。[70][71]而阻止轉運RNA與核醣體亞基上的信使RNA結合,表示胺基酸無法添加到多肽鏈中,也無法產生新的蛋白質,而阻止病菌生長,讓人體免疫系統有時間殺死和清除病菌。[72]
藥物動力學
[編輯]藥物幾乎完全經由服用者的小腸上部吸收。它在血漿中的最大血藥濃度於給藥後一到兩小時內發生。血漿蛋白結合率高達近80-90%。
獨克士黴素在人體的代謝微小,可忽略不計。經人體主動排泄到腸道(部分通過膽囊,部分經由血管),其中一些會形成螯合物而失去活性。有約40%經由腎臟消除,
歷史
[編輯]由於青黴素出現,在第二次世界大戰期間徹底改變病菌感染的治療方法,許多藥廠開始利用生物勘探以發現抗生素的模式。美國氰胺製藥(於2009年為輝瑞製藥併入)為其中之一,該公司的研究人員在1940年代末期發現金黴素,此為四環素類抗生素的第一種。[4]
隨後輝瑞製藥發明土黴素及四環黴素,最終製造出獨克士黴素,並於1960年代初進行臨床開發,於1967年獲得FDA批准用於醫療用途。[4]
當獨克士黴素的專利在1970年代初接近到期時,此專利成為輝瑞製藥和國際整流器公司[73]之間訴訟的主題,最終是輝瑞製藥勝訴。爭端直到1983年才落幕。[74]
FDA於2013年1月報告稱,"由於需求增加和製造問題" ,部分(並非全部)獨克士黴素出現短缺現象。[75]導致美國獨克士黴素的市場價格於2013年和2014年初大幅上漲(從每瓶500片的20美元上漲到超過1,800美元),[76][77][78]過後才下降。.[79][80]
社會與文化
[編輯]獨克士黴素在全球銷售,有多種商業品牌。[81]市面上也有其通用名藥物流通。[1][10]
研究
[編輯]有研究以此藥物應用於下述疾病的治療:

抗發炎劑
[編輯]一些研究顯示獨克士黴素是一可透過抑制促炎性細胞因子來發揮抗發炎特性的潛在藥劑。[84]
傷口癒合
[編輯]科學家們於近年積極投入研究,以開發出更有效的獨克士黴素,加速傷口癒合。研究的重點主要放在兩方面:
- 延長藥物保存期限
- 改善藥物投遞方式
最常見的投遞方式是傷口敷料。相較於過去單層的敷料,最新的設計已發展出三層結構,這種設計能更有效地將藥物釋放到傷口,促進癒合。[85]
研究試劑
[編輯]獨克士黴素和其他四環素類抗生素常被用作in vitro(體外)和in vivo(體內)生物醫學研究實驗中的研究試劑。
- 涉及細菌的實驗: 獨克士黴素和其他四環素類抗生素常被用於研究細菌相關的生物醫學實驗。
- 涉及真核細胞的實驗: 在研究真核細胞時,四環素系統常被用來控制基因的表達。研究人員藉由獨克士黴素可精確控制目標蛋白的產生時間和數量。
獨克士黴素在亞抗菌劑量下是基質金屬蛋白酶的抑制劑,並已為此目的而用於各種實驗,例如治療頑固性復發性角膜糜爛。[86]
參考文獻
[編輯]- ^ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 Doxycycline calcium. The American Society of Health-System Pharmacists. [2015-08-18]. (原始內容存檔於2015-09-23).
- ^ Malaria. Fit for Travel. Public Health Scotland. Chemoprophylaxis. [2023-12-04]. (原始內容存檔於2023-12-04).
- ^ Choosing a Drug to Prevent Malaria. Centers for Disease Control and Prevention. Doxycycline. February 2023 [2023-12-04]. (原始內容存檔於2016-11-13).
- ^ 4.0 4.1 4.2 Nelson ML, Levy SB. The history of the tetracyclines. Annals of the New York Academy of Sciences. December 2011, 1241 (1): 17–32. Bibcode:2011NYASA1241...17N. PMID 22191524. S2CID 34647314. doi:10.1111/j.1749-6632.2011.06354.x.
- ^ McFadden GI. Apicoplast. Current Biology. March 2014, 24 (7): R262–3. Bibcode:2014CBio...24.R262M. PMID 24698369. doi:10.1016/j.cub.2014.01.024
 .
.
- ^ Schlagenhauf-Lawlor P. Travelers' Malaria. PMPH-USA. 2008: 148. ISBN 978-1-55009-336-0.
- ^ Fischer J, Ganellin CR. Analogue-based Drug Discovery. John Wiley & Sons. 2006: 489. ISBN 978-3-527-60749-5.
- ^ Corey EJ. Drug discovery practices, processes, and perspectives. Hoboken, N.J.: John Wiley & Sons. 2013: 406 [2017-09-09]. ISBN 978-1-118-35446-9. (原始內容存檔於2023-01-14).
- ^ World Health Organization. World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ 10.0 10.1 Hamilton RJ. Tarascon pharmacopoeia 12th. Sudbury, MA: Jones & Bartlett Learning. 2011: 79. ISBN 978-1-4496-0067-9.
- ^ The Top 300 of 2022. ClinCalc. [2024-08-30]. (原始內容存檔於2024-08-30).
- ^ Doxycycline Drug Usage Statistics, United States, 2013 - 2022. ClinCalc. [2024-08-30]. (原始內容存檔於2020-07-08).
- ^ Sweet RL, Schachter J, Landers DV, Ohm-Smith M, Robbie MO. Treatment of hospitalized patients with acute pelvic inflammatory disease: comparison of cefotetan plus doxycycline and cefoxitin plus doxycycline. American Journal of Obstetrics and Gynecology. March 1988, 158 (3 Pt 2): 736–41. PMID 3162653. doi:10.1016/S0002-9378(16)44537-0.
- ^ Gjønnaess H, Holten E. Doxycycline (Vibramycin) in pelvic inflammatory disease. Acta Obstetricia et Gynecologica Scandinavica. 1978, 57 (2): 137–9. PMID 345730. S2CID 28328073. doi:10.3109/00016347809155893.
- ^ 15.0 15.1 Holmes NE, Charles PG. Safety and Efficacy Review of Doxycycline. Clinical Medicine. Therapeutics. 5 January 2009, 1: CMT.S2035. S2CID 58790579. doi:10.4137/CMT.S2035.
- ^ Määttä M, Kari O, Tervahartiala T, Peltonen S, Kari M, Saari M, Sorsa T. Tear fluid levels of MMP-8 are elevated in ocular rosacea--treatment effect of oral doxycycline. Graefe's Archive for Clinical and Experimental Ophthalmology = Albrecht von Graefes Archiv für Klinische und Experimentelle Ophthalmologie. August 2006, 244 (8): 957–62. PMID 16411105. S2CID 20540747. doi:10.1007/s00417-005-0212-3.
- ^ Quarterman MJ, Johnson DW, Abele DC, Lesher JL, Hull DS, Davis LS. Ocular rosacea. Signs, symptoms, and tear studies before and after treatment with doxycycline. Archives of Dermatology. January 1997, 133 (1): 49–54. PMID 9006372. doi:10.1001/archderm.133.1.49.
- ^ Walker DH, Paddock CD, Dumler JS. Emerging and re-emerging tick-transmitted rickettsial and ehrlichial infections. The Medical Clinics of North America. November 2008, 92 (6): 1345–61, x. PMID 19061755. doi:10.1016/j.mcna.2008.06.002.
- ^ Lee JJ, Chien AL. Rosacea in Older Adults and Pharmacologic Treatments. Drugs Aging. April 2024, 41 (5): 407–421. PMID 38649625. doi:10.1007/s40266-024-01115-y.
- ^ Rekart ML. Doxycycline:" New" treatment of choice for genital chlamydia infections.. British Columbia Medical Journal. December 2004, 46 (10): 503. (原始內容存檔於2017-02-02).
- ^ Doxycycline spectrum of bacterial susceptibility and Resistance (PDF). [2012-05-04]. (原始內容 (PDF)存檔於2014-02-01).
- ^ Stoddard RA, Galloway RL, Guerra MA. Leptospirosis. Yellow Book. Atlanta, GA: Centers for Disease Control and Prevention. 2015-07-10 [2017-04-16]. (原始內容存檔於2017-04-09).
- ^ Nadelman RB, Luger SW, Frank E, Wisniewski M, Collins JJ, Wormser GP. Comparison of cefuroxime axetil and doxycycline in the treatment of early Lyme disease. Annals of Internal Medicine. August 1992, 117 (4): 273–80. PMID 1637021. S2CID 23358315. doi:10.7326/0003-4819-117-4-273.
- ^ Luger SW, Paparone P, Wormser GP, Nadelman RB, Grunwaldt E, Gomez G, Wisniewski M, Collins JJ. Comparison of cefuroxime axetil and doxycycline in treatment of patients with early Lyme disease associated with erythema migrans. Antimicrobial Agents and Chemotherapy. March 1995, 39 (3): 661–7. PMC 162601
 . PMID 7793869. doi:10.1128/AAC.39.3.661.
. PMID 7793869. doi:10.1128/AAC.39.3.661.
- ^ Nadelman RB, Nowakowski J, Fish D, Falco RC, Freeman K, McKenna D, Welch P, Marcus R, Agüero-Rosenfeld ME, Dennis DT, Wormser GP. Prophylaxis with single-dose doxycycline for the prevention of Lyme disease after an Ixodes scapularis tick bite. The New England Journal of Medicine. July 2001, 345 (2): 79–84. PMID 11450675. doi:10.1056/NEJM200107123450201
 . 已忽略未知參數
. 已忽略未知參數|df=(幫助) - ^ Karlsson M, Hammers-Berggren S, Lindquist L, Stiernstedt G, Svenungsson B. Comparison of intravenous penicillin G and oral doxycycline for treatment of Lyme neuroborreliosis. Neurology. July 1994, 44 (7): 1203–7. PMID 8035916. S2CID 38661885. doi:10.1212/WNL.44.7.1203.
- ^ Weinstein RS. Human ehrlichiosis. American Family Physician. November 1996, 54 (6): 1971–6. PMID 8900357.
- ^ Karlsson U, Bjöersdorff A, Massung RF, Christensson B. Human granulocytic ehrlichiosis--a clinical case in Scandinavia. Scandinavian Journal of Infectious Diseases. 2001, 33 (1): 73–4. PMID 11234985. S2CID 218880245. doi:10.1080/003655401750064130.
- ^ 29.00 29.01 29.02 29.03 29.04 29.05 29.06 29.07 29.08 29.09 29.10 29.11 29.12 29.13 29.14 29.15 29.16 29.17 29.18 29.19 29.20 29.21 29.22 29.23 29.24 29.25 29.26 29.27 29.28 29.29 29.30 29.31 29.32 Doxycycline, ANDA no. 065055 Label. (PDF). U.S. Food and Drug Administration. 2012-12-14. (原始內容 (PDF)存檔於2014-04-19).
- ^ 30.00 30.01 30.02 30.03 30.04 30.05 30.06 30.07 30.08 30.09 30.10 30.11 30.12 30.13 30.14 30.15 30.16 30.17 30.18 30.19 30.20 30.21 30.22 30.23 30.24 30.25 30.26 30.27 30.28 30.29 30.30 30.31 Doxycycline, ANDA no. 065454 Label (PDF). U.S. Food and Drug Administration. 2008-07-16. (原始內容 (PDF)存檔於2013-10-19).
- ^ Gupta N, Boodman C, Jouego CG, Van Den Broucke S. Doxycycline vs azithromycin in patients with scrub typhus: a systematic review of literature and meta-analysis. BMC Infect Dis. December 2023, 23 (1): 884. PMC 10726538
 . PMID 38110855. doi:10.1186/s12879-023-08893-7
. PMID 38110855. doi:10.1186/s12879-023-08893-7  .
.
- ^ Anderson A, Bijlmer H, Fournier PE, Graves S, Hartzell J, Kersh GJ, Limonard G, Marrie TJ, Massung RF, McQuiston JH, Nicholson WL, Paddock CD, Sexton DJ. Diagnosis and management of Q fever--United States, 2013: recommendations from CDC and the Q Fever Working Group. MMWR. Recommendations and Reports. March 2013, 62 (RR-03): 1–30. PMID 23535757. (原始內容存檔於2014-04-19). 已忽略未知參數
|df=(幫助) - ^ Biggs HM, Behravesh CB, Bradley KK, Dahlgren FS, Drexler NA, Dumler JS, Folk SM, Kato CY, Lash RR, Levin ML, Massung RF, Nadelman RB, Nicholson WL, Paddock CD, Pritt BS, Traeger MS. Diagnosis and Management of Tickborne Rickettsial Diseases: Rocky Mountain Spotted Fever and Other Spotted Fever Group Rickettsioses, Ehrlichioses, and Anaplasmosis — United States. MMWR. Recommendations and Reports. 2016, 65 (2): 1–44 [2024-02-05]. PMID 27172113. doi:10.15585/mmwr.rr6502a1
 . (原始內容存檔於2023-01-28).
. (原始內容存檔於2023-01-28).
- ^ Schutze GE, Regan J, Bradley J. Use doxycycline as first-line treatment for rickettsial diseases. AAP News. American Academy of Pediatrics. 2010-07-01 [2024-02-05]. ISSN 1556-3332. (原始內容存檔於2024-02-05).
- ^ Spotted fever group rickettsial disease | DermNet. 2023-10-26 [2024-02-05]. (原始內容存檔於2024-02-05).
- ^ Okada T, Morozumi M, Tajima T, Hasegawa M, Sakata H, Ohnari S, Chiba N, Iwata S, Ubukata K. Rapid effectiveness of minocycline or doxycycline against macrolide-resistant Mycoplasma pneumoniae infection in a 2011 outbreak among Japanese children. Clinical Infectious Diseases. December 2012, 55 (12): 1642–9. PMID 22972867. doi:10.1093/cid/cis784.
- ^ Lyme disease. Treatment. 2018-12-21. (原始內容存檔於2016-06-10).
- ^ Taylor-Salmon E, Shapiro ED. Tick-borne infections in children in North America. Curr Opin Pediatr. April 2024, 36 (2): 156–163. PMC 10932821
 . PMID 38167816. doi:10.1097/MOP.0000000000001326. 已忽略未知參數
. PMID 38167816. doi:10.1097/MOP.0000000000001326. 已忽略未知參數|pmc-embargo-date=(幫助) - ^ Dreno B, Thiboutot D, Gollnick H, Bettoli V, Kang S, Leyden JJ, Shalita A, Torres V. Antibiotic stewardship in dermatology: limiting antibiotic use in acne. European Journal of Dermatology. 2014, 24 (3): 330–4. PMID 24721547. S2CID 28700961. doi:10.1684/ejd.2014.2309.
- ^ Garner SE, Eady A, Bennett C, Newton JN, Thomas K, Popescu CM. Minocycline for acne vulgaris: efficacy and safety. The Cochrane Database of Systematic Reviews. August 2012, 2012 (8): CD002086. PMC 7017847
 . PMID 22895927. doi:10.1002/14651858.CD002086.pub2. 已忽略未知參數
. PMID 22895927. doi:10.1002/14651858.CD002086.pub2. 已忽略未知參數|collaboration=(幫助) - ^ van Zuuren EJ, Fedorowicz Z, Carter B, van der Linden MM, Charland L. Interventions for rosacea. The Cochrane Database of Systematic Reviews. April 2015, 2015 (4): CD003262. PMC 6481562
 . PMID 25919144. doi:10.1002/14651858.CD003262.pub5. 已忽略未知參數
. PMID 25919144. doi:10.1002/14651858.CD003262.pub5. 已忽略未知參數|collaboration=(幫助) - ^ Cao H, Yang G, Wang Y, Liu JP, Smith CA, Luo H, Liu Y. Complementary therapies for acne vulgaris. The Cochrane Database of Systematic Reviews. January 2015, 1 (1): CD009436. PMC 4486007
 . PMID 25597924. doi:10.1002/14651858.CD009436.pub2. 已忽略未知參數
. PMID 25597924. doi:10.1002/14651858.CD009436.pub2. 已忽略未知參數|collaboration=(幫助) - ^ 43.0 43.1 Doxycycline Dosage Guide + Max Dose, Adjustments. [2024-02-05]. (原始內容存檔於2024-02-05).
- ^ Vanbaelen T, Manoharan-Basil SS, Kenyon C. 45 years of tetracycline post exposure prophylaxis for STIs and the risk of tetracycline resistance: a systematic review and meta-analysis. BMC Infect Dis. April 2024, 24 (1): 376. PMC 10996150
 . PMID 38575877. doi:10.1186/s12879-024-09275-3
. PMID 38575877. doi:10.1186/s12879-024-09275-3  .
.
- ^ Samuel K. Using antibiotics to prevent STIs. Aidsmap. 2023-05-26 [2024-03-10]. (原始內容存檔於2024-03-10).
- ^ Stewart J. Doxycycline Prophylaxis to Prevent Sexually Transmitted Infections in Women. New England Journal of Medicine. 2023-12-21, 389 (25): 2331–2340. PMC 10805625
 . PMID 38118022. doi:10.1056/NEJMoa2304007.
. PMID 38118022. doi:10.1056/NEJMoa2304007.
- ^ Cornelisse VJ, Riley B, Medland NA. Australian consensus statement on doxycycline post-exposure prophylaxis (doxy-PEP) for the prevention of syphilis, chlamydia and gonorrhoea among gay, bisexual and other men who have sex with men. Med J Aust. April 2024, 220 (7): 381–386. PMID 38479437. doi:10.5694/mja2.52258
 .
.
- ^ Guidelines for the Use of Doxycycline Post-Exposure Prophylaxis for Bacterial STI Prevention. CDC. 2023-09-29 [2024-03-10]. (原始內容存檔於2024-03-10).
- ^ 2023 Consensus Statement on doxycycline prophylaxis (Doxy-PEP) for the prevention of syphilis, chlamydia and gonorrhoea among gay, bisexual, and other men who have sex with men in Australia. Australasian Society for HIV Medicine. [10 March 2024]. (原始內容存檔於2024-03-10).
- ^ Highleyman L. Sexually transmitted infections in San Francisco have fallen since doxyPEP roll-out. Aidsmap. 5 March 2024 [2024-03-10]. (原始內容存檔於2024-03-10).
- ^ Hashemi SH, Gachkar L, Keramat F, Mamani M, Hajilooi M, Janbakhsh A, Majzoobi MM, Mahjub H. Comparison of doxycycline-streptomycin, doxycycline-rifampin, and ofloxacin-rifampin in the treatment of brucellosis: a randomized clinical trial. International Journal of Infectious Diseases. April 2012, 16 (4): e247–e251 [2014-08-23]. PMID 22296864. doi:10.1016/j.ijid.2011.12.003
 . (原始內容存檔於2021-08-28).
. (原始內容存檔於2021-08-28).
- ^ Doryx- doxycycline hyclate tablet, delayed release. DailyMed. 2020-10-23 [2022-03-05]. (原始內容存檔於2022-01-03).
- ^ Dahl EL, Shock JL, Shenai BR, Gut J, DeRisi JL, Rosenthal PJ. Tetracyclines specifically target the apicoplast of the malaria parasite Plasmodium falciparum. Antimicrobial Agents and Chemotherapy. September 2006, 50 (9): 3124–31. PMC 1563505
 . PMID 16940111. doi:10.1128/AAC.00394-06.
. PMID 16940111. doi:10.1128/AAC.00394-06.
- ^ Holmes NE, Charles PG. Safety and Efficacy Review of Doxycycline. Clinical Medicine. Therapeutics. 2009, 1: CMT.S2035. doi:10.4137/CMT.S2035.
- ^ Gaillard T, Madamet M, Pradines B. Tetracyclines in malaria. Malar J. November 2015, 14: 445. PMC 4641395
 . PMID 26555664. doi:10.1186/s12936-015-0980-0
. PMID 26555664. doi:10.1186/s12936-015-0980-0  .
.
- ^ Guidelines for the treatment of malaria. Geneva: World Health Organization. 2015: 246. ISBN 978-92-4-154912-7.
- ^ Hoerauf A, Mand S, Fischer K, Kruppa T, Marfo-Debrekyei Y, Debrah AY, Pfarr KM, Adjei O, Büttner DW. Doxycycline as a novel strategy against bancroftian filariasis-depletion of Wolbachia endosymbionts from Wuchereria bancrofti and stop of microfilaria production. Medical Microbiology and Immunology. November 2003, 192 (4): 211–6. PMID 12684759. S2CID 23349595. doi:10.1007/s00430-002-0174-6.
- ^ Taylor MJ, Makunde WH, McGarry HF, Turner JD, Mand S, Hoerauf A. Macrofilaricidal activity after doxycycline treatment of Wuchereria bancrofti: a double-blind, randomised placebo-controlled trial. Lancet. 2005, 365 (9477): 2116–21. PMID 15964448. S2CID 21382828. doi:10.1016/S0140-6736(05)66591-9.
- ^ Kaufman JA, Lee MJ. Vascular and interventional radiology 2nd. Philadelphia, PA: Saunders. 22 June 2013. ISBN 978-0-323-07672-2. OCLC 853455295.
- ^ Müller M, Butler J, Heidecker B. Emerging therapies in transthyretin amyloidosis – a new wave of hope after years of stagnancy?. European Journal of Heart Failure (Wiley). 7 January 2020, 22 (1): 39–53. PMID 31912620. doi:10.1002/ejhf.1695
 .
.
- ^ 61.0 61.1 Haberfeld H (編). Austria-Codex. Vienna: Österreichischer Apothekerverlag. 2020. Doxycyclin Genericon 200 mg lösliche Tabletten (德語).
- ^ ((PubMed Health)). Doxycycline (By mouth). U.S. National Library of Medicine. 2016-07-01 [2016-07-16]. (原始內容存檔於2013-11-11).
- ^ Chung AM, Reed MD, Blumer JL. Antibiotics and breast-feeding: a critical review of the literature. Paediatric Drugs. 2002, 4 (12): 817–37. PMID 12431134. S2CID 8595370. doi:10.2165/00128072-200204120-00006.
- ^ Mylonas I. Antibiotic chemotherapy during pregnancy and lactation period: aspects for consideration. Archives of Gynecology and Obstetrics. January 2011, 283 (1): 7–18. PMID 20814687. S2CID 25492353. doi:10.1007/s00404-010-1646-3.
- ^ 65.0 65.1 Side effects of doxycycline. NHSP. [2024-11-11].
- ^ Archer JS, Archer DF. Oral contraceptive efficacy and antibiotic interaction: a myth debunked. Journal of the American Academy of Dermatology. June 2002, 46 (6): 917–23. PMID 12063491. doi:10.1067/mjd.2002.120448.
- ^ Dréno B, Bettoli V, Ochsendorf F, Layton A, Mobacken H, Degreef H. European recommendations on the use of oral antibiotics for acne (PDF). European Journal of Dermatology. November–December 2004, 14 (6): 391–9. PMID 15564203.[永久失效連結]
- ^ DeRossi SS, Hersh EV. Antibiotics and oral contraceptives. Dental Clinics of North America. October 2002, 46 (4): 653–64. CiteSeerX 10.1.1.620.9933
 . PMID 12436822. doi:10.1016/S0011-8532(02)00017-4.
. PMID 12436822. doi:10.1016/S0011-8532(02)00017-4.
- ^ Flower R, Rang HP, Dale MM, Ritter JM, Henderson G. Rang & Dale's Pharmacology. Edinburgh: Churchill Livingstone. 2012. ISBN 978-0-7020-3471-8.
- ^ Hitchings A, Lonsdale D, Burrage D, Baker E. Top 100 drugs: clinical pharmacology and practical prescribing. Churchill Livingstone. 2015: 200–201. ISBN 978-0-7020-5516-4.
- ^ Maaland MG, Papich MG, Turnidge J, Guardabassi L. Pharmacodynamics of doxycycline and tetracycline against Staphylococcus pseudintermedius: proposal of canine-specific breakpoints for doxycycline. Journal of Clinical Microbiology. November 2013, 51 (11): 3547–54. PMC 3889732
 . PMID 23966509. doi:10.1128/JCM.01498-13.
. PMID 23966509. doi:10.1128/JCM.01498-13.
- ^ Doxycycline. www.drugbank.ca. [2019-01-23]. (原始內容存檔於2008-11-10).
- ^ Pfizer, Inc. v. International Rectifier Corp., 545 F. Supp. 486 (C.D. Cal. 1980). Justia Law. (原始內容存檔於2015-02-24).
- ^ Pfizer to Get Rachelle Units. The New York Times. The Associated Press. 1983-07-06. (原始內容存檔於2016-03-06).
- ^ Nationwide Shortage of Doxycycline: Resources for Providers and Recommendations for Patient Care. CDC Health Alert Network. 2013-06-12. (原始內容存檔於2015-02-15).
- ^ Sudden increase in cost of common drug concerns many. WSMV-TV. 2013-03-12. (原始內容存檔於2014-12-31).
- ^ Rosenthal E. Officials Question the Rising Costs of Generic Drugs. The New York Times. 2014-10-07. (原始內容存檔於2017-02-23).
- ^ Palmer E. Hikma hits the jackpot with doxycycline shortage. FiercePharmaManufacturing. 2014-03-13. (原始內容存檔於2015-01-01).
- ^ Costco Drug Information. [2016-07-31]. (原始內容存檔於2016-03-04).
- ^ Doxycycline Hyclate Prices and Doxycycline Hyclate Coupons. GoodRx. [2016-07-31]. (原始內容存檔於2016-07-28).
- ^ International availability for doxycycline. drugs.com. [2015-04-29]. (原始內容存檔於2015-05-16).
- ^ Leung E, Landa G. Update on current and future novel therapies for dry age-related macular degeneration. Expert Review of Clinical Pharmacology. September 2013, 6 (5): 565–79. PMID 23971874. S2CID 26680094. doi:10.1586/17512433.2013.829645.
- ^ Greenwald RA. The road forward: the scientific basis for tetracycline treatment of arthritic disorders. Pharmacological Research. December 2011, 64 (6): 610–3. PMID 21723947. doi:10.1016/j.phrs.2011.06.010.
- ^ Krakauer, Teresa; Buckley, Marilyn. Doxycycline Is Anti-Inflammatory and Inhibits Staphylococcal Exotoxin-Induced Cytokines and Chemokines. Antimicrobial Agents and Chemotherapy. November 2003, 47 (11): 3630–3633 [2024-11-11]. doi:10.1128/AAC.47.11.3630-3633.2003.
- ^ Saliy O, Popova M, Tarasenko H, Getalo O. Development strategy of novel drug formulations for the delivery of doxycycline in the treatment of wounds of various etiologies. Eur J Pharm Sci. April 2024, 195: 106636. PMID 38185273. doi:10.1016/j.ejps.2023.106636
 .
.
- ^ Dursun D, Kim MC, Solomon A, Pflugfelder SC. Treatment of recalcitrant recurrent corneal erosions with inhibitors of matrix metalloproteinase-9, doxycycline and corticosteroids. American Journal of Ophthalmology. July 2001, 132 (1): 8–13. PMID 11438047. doi:10.1016/S0002-9394(01)00913-8.
